drawing the outer electron box diagram of a transition metal Draw an outer electron box diagram for a Tc cation. A + 3 cation of a certain transition metal has two electrons in its outermost d subshell. Which transition metal could this be?
As long as you have the required conductor length inside the old panel (you will find you have more than plenty as minimum) you can use it as a junction box after you gut the .
0 · transition metal cations diagram
1 · orbital electron structure diagram
2 · orbital electron configuration diagram
3 · orbital diagram of transition metals
4 · orbital box notation of electrons
5 · electron configuration in orbital box
6 · electron configuration diagram pdf
7 · 40 electron configuration diagram
Item may be available in your local warehouse, prices may vary. This product is expected to be in stock and available for purchase soon. Check back again later. Your delivery date is not .
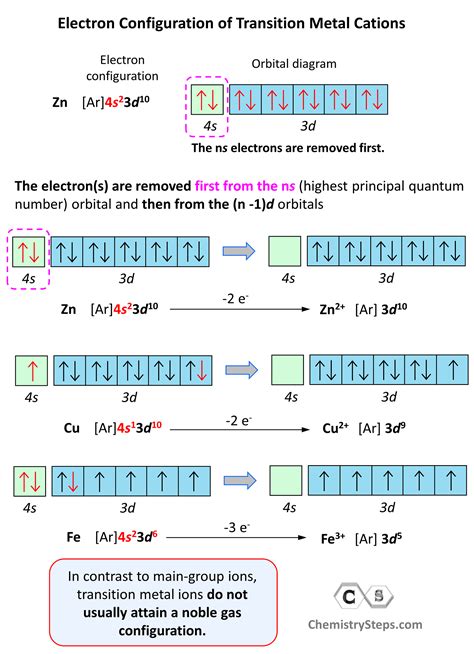
The electron configuration for the first row transition metals consists of 4s and 3d subshells with an argon (noble gas) core. This only applies to the first row transition metals, adjustments will be necessary when writing .Question: Drawing the outer electron box diagram of a transition metal cation Draw an outer electron box diagram for a Zrcation. Show transcribed image text There are 2 steps to solve this one. Writing an electron configuration for a transition metal ion starts with the same steps as writing the configuration of an s- or p-block cation: Write the electron configuration for the neutral atom and then determine the number .Our expert help has broken down your problem into an easy-to-learn solution you can count on. Question: Drawing the outer electron box diagram of a transition metal c Draw an outer electron box diagram for a Mn3 cation. Here’s the best .
transition metal cations diagram
orbital electron structure diagram
Transition elements or transition metals. These are metallic elements in which the last electron added enters a d orbital. The valence electrons (those added after the last noble gas configuration) in these elements include the ns and ( n – 1) .Draw an outer electron box diagram for a Tc cation. A + 3 cation of a certain transition metal has two electrons in its outermost d subshell. Which transition metal could this be?Electron configurations and orbital box diagrams can be written right from the periodic table. The periodic table below, shows the s, p, d, and f-blocks. When reading the periodic table from left to right, one can easily write an electron .
Study with Quizlet and memorize flashcards containing terms like Define a transition element, Draw the electron box diagram for chromium and copper, Why is scandium not a transition element? and more.
The electron configuration for the first row transition metals consists of 4s and 3d subshells with an argon (noble gas) core. This only applies to the first row transition metals, adjustments will be necessary when writing the electron configuration for the other rows of transition metals.Electron Configuration of d Elements – the Transition Series. There is one exception to keep in mind for the electron configuration of transition metals. That is the (n +1)s orbitals always fill before the nd orbitals.Question: Drawing the outer electron box diagram of a transition metal cation Draw an outer electron box diagram for a Zrcation. Show transcribed image text There are 2 steps to solve this one.
metal fabrication shops in maine
orbital electron configuration diagram
metal fabrication shops in houston tx
The periodic table can be divided into three categories based on the orbital in which the last electron to be added is placed: main group elements (s and p orbitals), transition elements (d orbitals), and inner transition elements (f orbitals). Writing an electron configuration for a transition metal ion starts with the same steps as writing the configuration of an s- or p-block cation: Write the electron configuration for the neutral atom and then determine the number of electrons that are lost to form the cation.Our expert help has broken down your problem into an easy-to-learn solution you can count on. Question: Drawing the outer electron box diagram of a transition metal c Draw an outer electron box diagram for a Mn3 cation. Here’s the best way to solve it.Transition elements or transition metals. These are metallic elements in which the last electron added enters a d orbital. The valence electrons (those added after the last noble gas configuration) in these elements include the ns and ( n – 1) d electrons.
Draw an outer electron box diagram for a Tc cation. A + 3 cation of a certain transition metal has two electrons in its outermost d subshell. Which transition metal could this be?
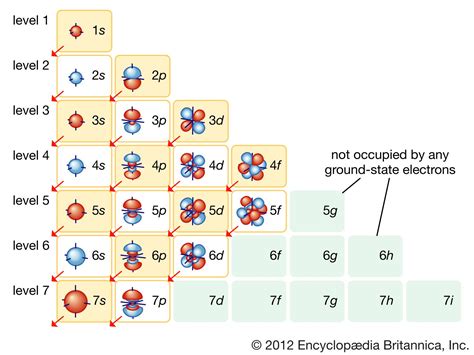
Electron configurations and orbital box diagrams can be written right from the periodic table. The periodic table below, shows the s, p, d, and f-blocks. When reading the periodic table from left to right, one can easily write an electron configuration without memorizing the filling order.Study with Quizlet and memorize flashcards containing terms like Define a transition element, Draw the electron box diagram for chromium and copper, Why is scandium not a transition element? and more. The electron configuration for the first row transition metals consists of 4s and 3d subshells with an argon (noble gas) core. This only applies to the first row transition metals, adjustments will be necessary when writing the electron configuration for the other rows of transition metals.
Electron Configuration of d Elements – the Transition Series. There is one exception to keep in mind for the electron configuration of transition metals. That is the (n +1)s orbitals always fill before the nd orbitals.Question: Drawing the outer electron box diagram of a transition metal cation Draw an outer electron box diagram for a Zrcation. Show transcribed image text There are 2 steps to solve this one.The periodic table can be divided into three categories based on the orbital in which the last electron to be added is placed: main group elements (s and p orbitals), transition elements (d orbitals), and inner transition elements (f orbitals).
orbital diagram of transition metals
Writing an electron configuration for a transition metal ion starts with the same steps as writing the configuration of an s- or p-block cation: Write the electron configuration for the neutral atom and then determine the number of electrons that are lost to form the cation.
Our expert help has broken down your problem into an easy-to-learn solution you can count on. Question: Drawing the outer electron box diagram of a transition metal c Draw an outer electron box diagram for a Mn3 cation. Here’s the best way to solve it.
Transition elements or transition metals. These are metallic elements in which the last electron added enters a d orbital. The valence electrons (those added after the last noble gas configuration) in these elements include the ns and ( n – 1) d electrons.
Draw an outer electron box diagram for a Tc cation. A + 3 cation of a certain transition metal has two electrons in its outermost d subshell. Which transition metal could this be?Electron configurations and orbital box diagrams can be written right from the periodic table. The periodic table below, shows the s, p, d, and f-blocks. When reading the periodic table from left to right, one can easily write an electron configuration without memorizing the filling order.
metal fabrication shops near franklin mass
Students will learn the required skills to operate safely in a machining .
drawing the outer electron box diagram of a transition metal|orbital electron structure diagram